Partnering & Collaboration
Together for better cancer diagnostics
Scientific Excellence
Our roots lie in cancer research, and our solutions are the result of rigorous scientific work. As a company offering services and solutions to improve cancer therapy decisions, we feel closely connected to research and committed to collaboration in scientific projects. We are actively engaged in various publicly funded projects.
Please reach out to us if you have project ideas where our expertise could be valuable.
OVARSENSE
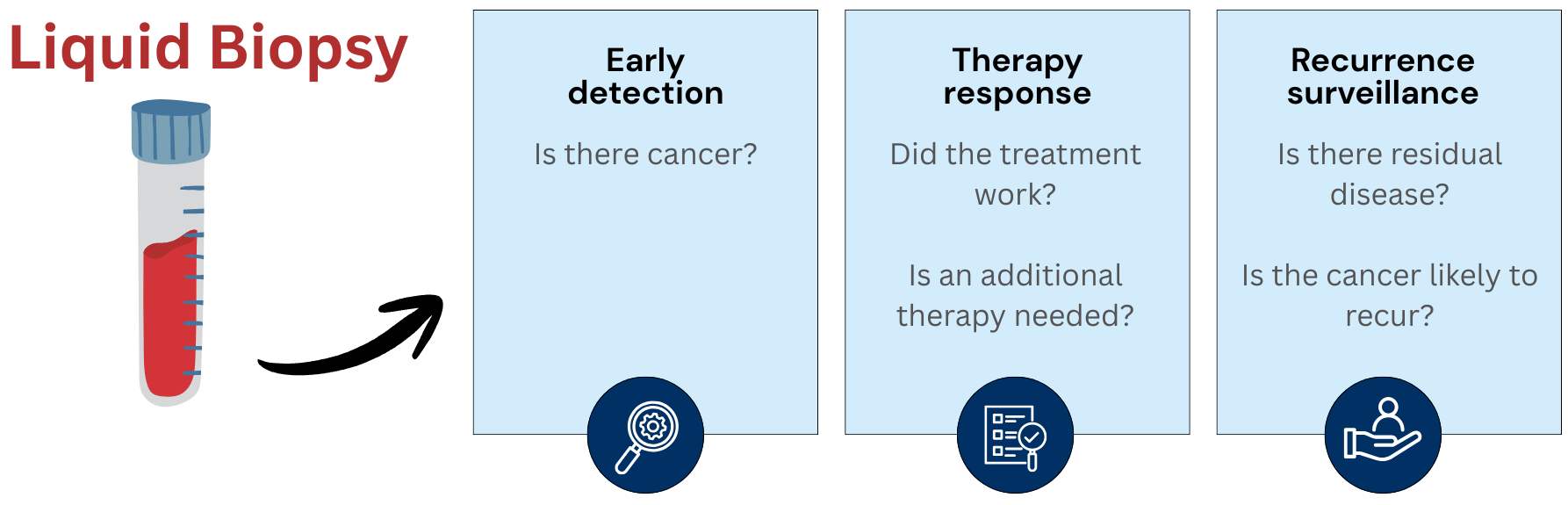
Ovarian cancer is often diagnosed at an advanced stage.
In the collaborative
project OVARSENSE, we aim to develop a test in which a simple blood sample (liquid biopsy) is sufficient to
detect ovarian cancer and potential recurrences during and after therapy (minimal residual disease, MRD) at an
earlier stage, thereby enabling more targeted treatment of ovarian cancer.
Funding

Cooperation Partners

Project goal
A test that can both determine minimal residual disease, as well as being used for the early detection of ovarian cancer is under development. This minimally invasive method reduces the burden on patients and offers significantly higher accuracy than traditional methods.
Approach
The goal is to enable precise and personalized diagnostics through the analysis of circulating tumor DNA (ctDNA) using liquid biopsies and next-generation sequencing (NGS). This innovative technology allows for the early detection of recurrences and supports personalized therapy decisions.
Project Consortium
- Liqomics GmbH Cologne, Germany
- Gynäkologisches Krebszentrum, Evangelisches Krankenhaus Bergisch Gladbach gGmbH Bergisch Gladbach, Germany
- Miltenyi Biotec B.V. & Co. KG Bergisch Gladbach, Germany
FOCuS
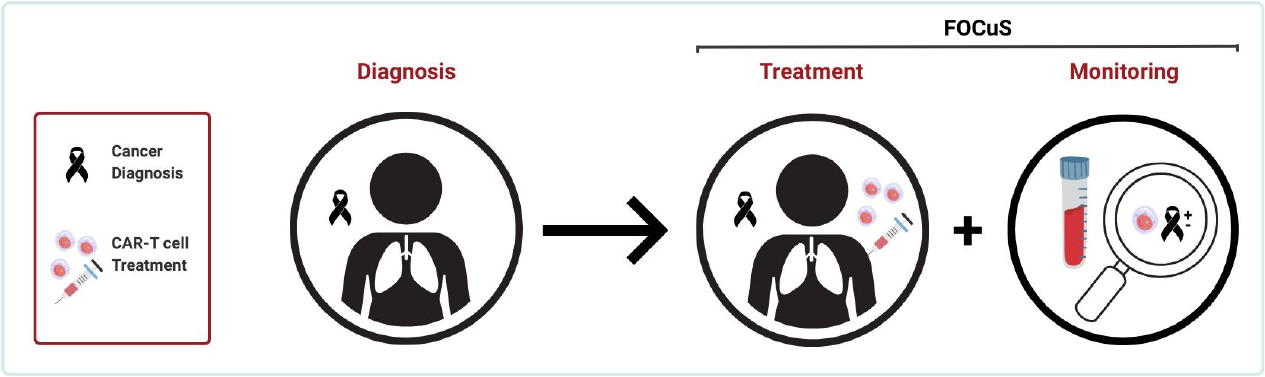
Small cell lung cancer (SCLC) is a type of tumor with an extremely low survival rate. In our collaborative project FOCuS, we aim to develop a new form of immune cell therapy together with a companion cancer test (liquid biopsy). The goal is to enable earlier, more targeted, and more effective treatment of SCLC through the combined development of therapy and diagnostic testing.
Funding

Cooperation Partners

Project Objective
The aim of the project is to develop a CAR T-cell therapy for the treatment of small cell lung cancer (SCLC).
Approach
We use non-viral genome editing with CRISPR/Cas9 for the targeted generation of optimized CKI CAR T cells. In addition, we are developing a liquid biopsy–based testing system that enables longitudinal molecular characterization of SCLC during therapy, including MRD monitoring and subtyping.
Project Consortium
- University Hospital Cologne (AöR) University Hospital Cologne (AöR)
- Liqomics GmbH Cologne, Germany
- Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. Munich, Germany
- Fraunhofer Institut für Zelltherapie und Immunologie, IZI Leipzig, Germany